the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
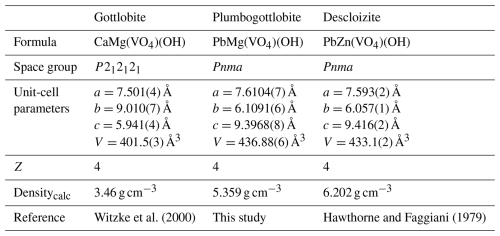
Plumbogottlobite, PbMg(VO4)(OH), the Pb analogue of gottlobite and the Mg analogue of descloizite
Joy Désor
Plumbogottlobite (IMA 2025-075), PbMg(VO4)(OH), is a new mineral from the Glücksstern mine, Gottlob Hill, Friedrichroda, Gotha District, Thuringia, Germany; it is a late-stage, low-temperature hydrothermal mineral occurring on baryte and hausmannite. Plumbogottlobite forms orange-brown tablets up to about 0.2 mm in diameter. The mineral has a light-orange-brown streak, adamantine lustre, a Mohs hardness of ∼ 4 to 4.5, brittle tenacity, irregular to conchoidal fracture, no cleavage, and a calculated density of 5.359 g cm−3. Optically, plumbogottlobite crystals are biaxial (–), with 2V=84(1)°. The empirical formula from electron probe microanalyses based on 5 O apfu is (Pb0.89□0.11)Σ1.00(Mg0.77CuMn)Σ1.05(VO4)(OH) (+0.12 H for charge balance). Plumbogottlobite is orthorhombic with space group Pnma and single-crystal unit-cell parameters a=7.6104(7), b=6.1091(6), c=9.3968(8) Å, V=436.88(7) Å3, and Z=4. The crystal structure is refined to R1=0.0302 for 429 reflections with I>2σI. The mineral is the Pb analogue of gottlobite and the Mg analogue of descloizite.
- Article
(3363 KB) - Full-text XML
-
Supplement
(348 KB) - BibTeX
- EndNote
Đorđević et al. (2016) noted 17 minerals and 12 synthetic compounds with the descloizite–adelite structure topology and provided the general formula M12(OH,O)[(O4,O3OH)], where M1 = Na+, Ca2+, Cd2+, Hg2+, Pb2+; M2 = Mg2+, Al3+, Mn, Fe2+, Co2+, Ni2+, Cu2+, Zn2+; and X = Si4+, P5+, V5+, As5+, Mo6+. Among the minerals are eight arsenates, seven vanadates, and two silicates. One of the vanadates, gottlobite, CaMg(VO4)(OH), has only been found at the Glücksstern mine, Gottlob Hill, Thuringia, Germany. In examining apparent gottlobite crystals from the Glücksstern mine, we found them to be the Pb analogue of the mineral, with the ideal formula PbMg(VO4)(OH).
The new mineral is named plumbogottlobite based on it being the Pb analogue of gottlobite (Witzke et al., 2000). Note that we chose this name rather than magnesiodescloizite because the mineral is virtually identical in appearance to gottlobite and occurs in the same general mineral assemblage at the Glücksstern mine. The mineral and its name have been approved by the IMA Commission on New Minerals, Nomenclature and Classification (CNMNC), proposal IMA2025-075 (Warr symbol: Pgot). The holotype specimen is deposited in the collections of the Natural History Museum of Los Angeles County, 900 Exposition Boulevard, Los Angeles, CA 90007, USA, catalogue number 77403.
Plumbogottlobite was found on the dumps of the Glücksstern mine, Gottlob Hill, Friedrichroda, Gotha District, Thuringia, Germany (50°51′6′′ N, 10°34′10′′ E). The Glücksstern mine was operated for manganese and iron until its closure in 1855 (Scheven, 1990). The mine is also the type locality for crednerite, CuMnO2 (Rammelsberg, 1849); gottlobite, CaMg(VO4)(OH) (Witzke et al., 2000); vésigniéite, BaCu3(VO4)2(OH)2 (Guillemin, 1955); and wakefieldite-(La) (Witzke et al., 2008). Plumbogottlobite is a late-stage, low-temperature hydrothermal mineral occurring on baryte and hausmannite.
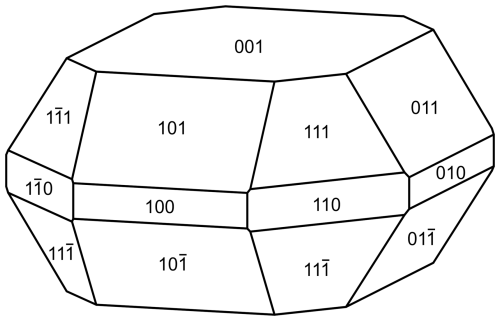
Plumbogottlobite occurs as tablets, up to about 0.2 mm in diameter (Fig. 1). The mineral is identical in appearance to gottlobite. The tablets are flattened on {001} and exhibit the forms {100}, {010}, {001}, {110}, {011}, {101}, and {111} (Fig. 2). No twinning was observed. The colour is orange-brown, and the streak is light orange-brown. The mineral has adamantine lustre and is transparent. No fluorescence was observed in either long- or short-wave ultraviolet illumination. The Mohs hardness is about 4 to 4.5 based on scratch tests. The tenacity is brittle, and the fracture is irregular to conchoidal. The density could not be measured because crystals exceed the density of available density fluids. The density calculated from the empirical formula and single-crystal unit cell is 5.359 g cm−3. At room temperature, plumbogottlobite rapidly decomposes in dilute HCl, forming a white residue, which slowly dissolves.

Figure 1Plumbogottlobite crystals on baryte and hausmannite on holotype specimen 77403; the field of view is 0.37 mm across.
Optically, plumbogottlobite crystals are uniaxial (–). The indices of refraction could not be measured because they are higher than available immersion liquids. The average index of refraction calculated from the Gladstone–Dale relationship (Mandarino, 1981) is 2.077. The 2V (meas.) was determined from extinction data, obtained on a spindle stage in a liquid of n=1.78 and analysed using EXCALIBRW (Gunter et al., 2004), is 84(1)°. The partially determined optical orientation is Y=c. No pleochroism was observed.
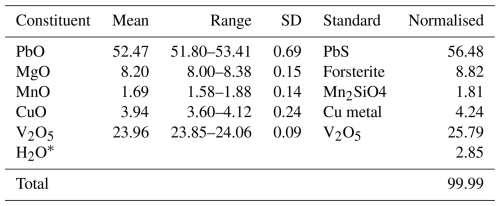
Crystals of plumbogottlobite were analysed at Caltech on a JEOL JXA-iHP200F field emission electron probe microanalyser (EPMA) in wavelength dispersive spectroscopy (WDS) mode (four points). The analytical conditions were 15 kV accelerating voltage, 10 nA beam current, and 5 µm beam diameter. Insufficient material is available for the determination of H2O, and so it was calculated based on the structure (V = 1 and O = 5). No other elements were detected in energy-dispersive spectroscopy (EDS) analyses. The crystals did not take a good polish, which provided analyses that are estimated to be low by about 7.5 %. Consequently, the analyses have been normalised. Analytical data are given in Table 1.
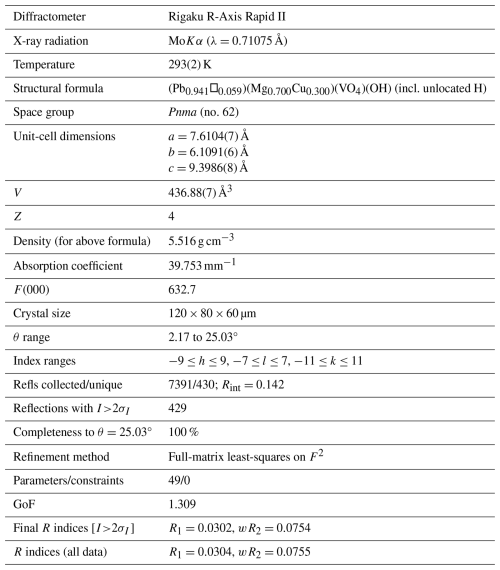
Table 3Data collection and structure refinement for plumbogottlobite.
Note that . GoF = S = . . wR2 = ; , where a is 0, b is 8.134, and P is .
The empirical formula (based on 5 O apfu), with cations allotted to structural sites, is (Pb0.89Mn)Σ1.00(Mg0.77CuMn)Σ1.00(VO4)(OH) (+0.12 H for charge balance). The simplified formula is (Pb,Mn,□)(Mg,Cu,Mn)(VO4)(OH), and the ideal formula is PbMg(VO4)(OH), which requires PbO 61.41, MgO 11.09, V2O5 25.02, and H2O 2.48, totalling 100 wt %.
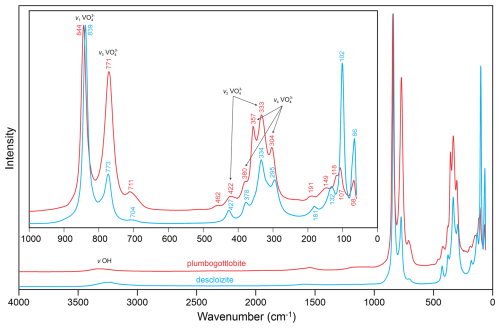
Raman spectroscopy was conducted on single-crystal fragments on a Horiba XploRA PLUS spectrometer using a 532 nm diode laser, 20 5 s accumulations at 2 mW power with a 100 µm slit, a 1800 grooves mm−1 diffraction grating, and a 100× (0.9 NA) objective. The spectrum of plumbogottlobite from 4000 to 60 cm−1 is compared with that of descloizite from Berg Aukas, Namibia (Natural History Museum of Los Angeles County, catalogue number 68303), in Fig. 3. Plumbogottlobite is very sensitive to the laser, requiring a relatively low power of 2 mW, whereas the descloizite spectrum was successfully recorded at 16 mW. The descloizite spectrum was chosen for comparison because of the mineral's similar composition, PbZn(VO4)(OH), and equivalent structure.
The spectrum of plumbogottlobite is very similar to that of descloizite. A broad, weak band is seen in both spectra in the OH stretching region. The very weak feature at about 1550 cm−1 is probably an overtone. Between 1000 and 200 cm−1, both spectra are dominated by bands related to the stretching and bending vibrations of the VO4 tetrahedron. The mode assignments shown in Fig. 3 are based on Frost et al. (2001), Martens et al. (2003), and Đorđević et al. (2016).
Table 4Refined atom coordinates, displacement parameters (Å2), and site occupancies for plumbogottlobite.
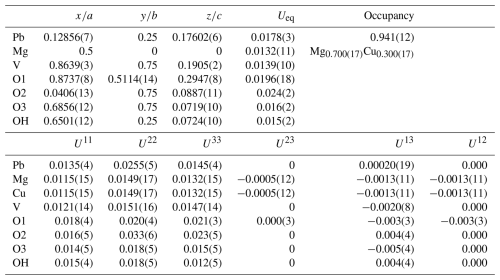
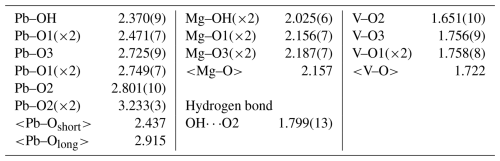
Table 6Bond valence analysis for plumbogottlobite. Values are expressed in valence units (vu).

Bond valence parameters are from Gagné and Hawthorne (2015). Hydrogen bond valence is based on the OO bond lengths from Ferraris and Ivaldi (1988); the negative value indicates donated bond valence.
6.1 X-ray powder diffraction
X-ray powder diffraction data were recorded using a Rigaku R-Axis Rapid II curved imaging plate microdiffractometer with monochromatised MoKα radiation. A Gandolfi-like motion on the ϕ and ω axes was used to randomise the sample. Observed d values and intensities were derived by profile fitting using JADE Pro software (Materials Data, Inc.). The calculated intensities were calculated from the structure, also using JADE Pro software. Data are given in Table 2. The unit-cell parameters refined from the powder data using JADE Pro with whole-pattern fitting (space group Pnma) are a=7.599(12), b=6.118(10), c=9.394(15) Å, and V=436.7(10) Å3.
6.2 Single-crystal diffraction
Single-crystal X-ray studies were also done using a Rigaku R-Axis Rapid II curved imaging plate microdiffractometer with monochromatised MoKα radiation. The Rigaku CrystalClear software package was used for processing the structure data, including the application of a numerical absorption correction. Note that an empirical absorption correction using the multi-scan method resulted in an almost identical Rint. The relatively high Rint is probably due to the presence of a small satellite crystal. Useful data could only be obtained out to θ=25°, which accounts for the relatively low data-to-parameter ratio. The structure was solved in space group Pnma using the intrinsic-phasing algorithm of SHELXT (Sheldrick, 2015a). SHELXL-2016 (Sheldrick, 2015b) was used for the refinement of the structure. We also attempted a structure solution and refinement in space group P212121, the space group reported for gottlobite by Đorđević et al. (2016). The refinement provided approximately the same final R1; however, the O4 site in that refinement could not be refined anisotropically without becoming non-positive definite (NPD), and checkcif provided a level-B alert, indicating that the correct space group was probably Pnma.
The structure solution in Pnma located all non-hydrogen sites, which were successfully refined with anisotropic displacement parameters. The occupancy of the Pb site was refined to 0.941(12). This compares reasonably well with the results of the EPMA, which provided 0.89 Pb apfu; however, because the EPMA showed an excess of 0.05 cations assignable to the Mg site, the possibility exists that a small amount of Mn2+ could be hosted in the Pb site. The refined site-scattering value of 308.64 e for the Pb site is consistent with the site occupancy (Pb0.93Mn0.05□0.02). The Mg site was refined with joint occupancy by Mg and Cu resulting in an occupancy of Mg0.700(17)Cu0.300(17) for a site-scattering value of 68.40 e. If 0.05 Mn apfu is assigned to the Pb site, the empirical formula based on the EPMA has (Mg0.77CuMn) in the Mg site, which yields a site-scattering value of 63.00e. This suggests that the structure crystal contains less Mg and more Cu and/or Mn than provided by the EPMA. For example, (Mg0.68CuMn) in the Mg site would provide a scattering value of 68.48 e. Efforts to locate the H site were unsuccessful. The data collection and refinement details are given in Table 3, atom coordinates and displacement parameters are given in Table 4, selected bond distances are given in Table 5, and a bond valence analysis is given in Table 6. The bond valence analysis (using the refined site occupancies) clearly indicates that OH is OH−, and all other O sites are O2−.
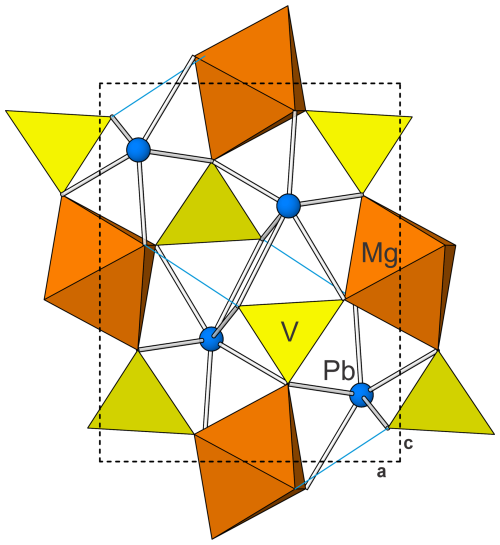
Plumbogottlobite has the same structure as descloizite, PbZn(VO4)(OH), and other minerals with the descloizite structure type. It is the Pb analogue of gottlobite and the Mg analogue of descloizite; selected data are presented in Table 7. As noted by Đorđević et al. (2016), gottlobite, CaMg(VO4)(OH), has an adelite-type structure with space group P212121, which is topologically equivalent to the descloizite structure. Đorđević et al. (2016) noted that most phases with the descloizite–adelite structure topology that have the descloizite structure type are lead vanadates.
In the structure of plumbogottlobite, edge-sharing chains of MgO4(OH)2 octahedra running parallel to [010] are corner-linked by VO4 tetrahedra to form a three-dimensional framework. Cavities within the framework host Pb2+ cations. The Pb2+ is nine-coordinated, with three short bonds (2.37–2.47 Å) on one side and six longer bonds (2.73–3.23 Å) on the other, clearly showing the 6s2 lone-pair electrons of Pb2+ to be stereoactive. The structure is shown in Fig. 4.
Crystallographic data for plumbogottlobite are available in the Supplement.
The supplement related to this article is available online at https://doi.org/10.5194/ejm-38-209-2026-supplement.
ARK oversaw the research, determined the physical and optical properties, did the Raman spectroscopy, did the X-ray diffraction studies, and wrote the paper. JD conducted initial characterisation studies on the mineral and identified it as a potential new species. CM did the electron probe microanalyses.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
The reviewers, Uwe Kolitsch and Jiří Sejkora, are thanked for their constructive comments on the paper.
The EPMA was carried out at the Caltech GPS Division Analytical Facility, which is supported, in part, by NSF grant no. EAR-2117942. A portion of this study was funded by the John Jago Trelawney Endowment to the Mineral Sciences Department of the Natural History Museum of Los Angeles County.
This paper was edited by Sergey Krivovichev and reviewed by Uwe Kolitsch and Jiří Sejkora.
Đorđević, T., Kolitsch, U., and Nasadala, L.: A single-crystal X-ray and Raman spectroscopic study of hydrothermally synthesized arsenates and vanadates with the descloizite and adelite structure types, Am. Mineral., 101, 1135–1149, 2016.
Ferraris, G. and Ivaldi, G.: Bond valence vs bond length in OO hydrogen bonds, Acta Crystallogr., B44, 341–344, 1988.
Frost, R. L., Williams, P. A., Kloprogge, J. T., and Leverett, P.: Raman spectroscopy of descloizite and mottramite at 298 and 77 K, J. Raman Spectrosc., 32, 906–911, 2001.
Gagné, O. C. and Hawthorne, F. C.: Comprehensive derivation of bond-valence parameters for ion pairs involving oxygen, Acta Crystallogr., B71, 562–578, 2015.
Guillemin, C.: Une nouvelle espèce minéral: la vésigniéite, Cu3Ba(VO4)2(OH)2, C. R. Hebd. Séances Acad. Sci., 240, 2331–2333, 1955.
Gunter, M. E., Bandli, B. R., Bloss, F. D., Evans, S. H., Su, S. C., and Weaver, R.: Results from a McCrone spindle stage short course, a new version of EXCALIBR, and how to build a spindle stage, The Microscope, 52, 23–39, 2004.
Hawthorne, F. C. and Faggiani, R.: Refinement of the structure of descloizite, Acta Crystallogr., B35, 717–720, 1979.
Mandarino, J. A.: The Gladstone-Dale relationship: Part IV. The compatibility concept and its application, Can. Mineral., 19, 441–450, 1981.
Martens, W., Frost, R. L., and Williams, P. A.: Molecular structure of the adelite group of minerals – a Raman spectroscopic study, J. Raman Spectrosc., 34, 104–111, 2003.
Rammelsberg, C.: Ueber das Mangankupfererz von Friedrichsrode, Zusatz zu der vorhergehenden Abhandlung, Poggendorffs Annalen der Physik und Chemie, 150, 559–562, 1849.
Scheven, M.: Die Minerale des Fundgebietes am Gottlob bei Friedrichroda im Thüringer Wald, Fundgrube, 26, 7–11, 1990.
Sheldrick, G. M.: SHELXT – Integrated space-group and crystal-structure determination, Acta Crystallogr., A71, 3–8, 2015a.
Sheldrick, G. M.: Crystal Structure refinement with SHELX, Acta Crystallogr., C71, 3–8, 2015b.
Witzke, T., Steins, M., Doering, T., and Kolitsch, U.: Gottlobite, CaMg(VO4,AsO4)(OH), a new mineral from Friedrichroda, Thuringia, Germany, N. Jb. Miner. Mon., 10, 444–454, 2000.
Witzke, T., Kolitsch, U., Warnsloh, J. M., and Göske, J.: Wakefieldite-(La), LaVO4, a new mineral species from the Glücksstern Mine, Friedrichroda, Thuringia, Germany, Eur. J. Mineral., 20, 1135–1139, 2008.
- Abstract
- Introduction
- Occurrence and associated minerals
- Physical and optical properties
- Chemical composition
- Raman spectroscopy
- Crystallography
- Description of the structure
- Data availability
- Author contributions
- Competing interests
- Disclaimer
- Acknowledgements
- Financial support
- Review statement
- References
- Supplement
- Abstract
- Introduction
- Occurrence and associated minerals
- Physical and optical properties
- Chemical composition
- Raman spectroscopy
- Crystallography
- Description of the structure
- Data availability
- Author contributions
- Competing interests
- Disclaimer
- Acknowledgements
- Financial support
- Review statement
- References
- Supplement