the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
When mineralogy meets the organic chemistry: delchiaroite, Cu3I(CH3S)2, the first copper iodide–methanethiolate from the Carrara marble quarries, Apuan Alps, Tuscany, Italy
Cristian Biagioni
Jiří Sejkora
Yves Moëlo
Antonino Criscuolo
Zdeněk Dolníček
The new mineral delchiaroite, Cu3I(CH3S)2, has been discovered in La Piana quarry, Colonnata marble basin, Carrara, Apuan Alps, Tuscany, Italy. It occurs as light-yellow acicular crystals, elongated on [010], up to 0.1 mm in length and is brittle, with a light-yellow streak and a greasy luster. In reflected light, delchiaroite is light gray with abundant internal yellow reflections; anisotropism is moderate, with gray tints. Reflectance values for the four COM wavelengths are (λ (nm) (%)) 470: 20.6/22.2; 546: 20.9/23.4; 589: 20.7/23.3; and 650: 20.1/22.4. Electron microprobe analysis resulted in the following (average of 12 spot analyses – in wt %): Cu 47.27, I 31.09, S 14.90, Ccalc 5.58, Hcalc 1.40, and total 100.24. The empirical formula of delchiaroite, calculated on the basis of Cu + I + S = 6 atoms per formula unit, is Cu3.07I1.01(CH3S)1.92, in accordance with the end-member formula Cu3I(CH3S)2. Delchiaroite is orthorhombic, space group Pmmn, with a=16.924(10), b=4.099(2), c=5.572(3) Å, V=386.5(4) Å3, and Z=2. The crystal structure was refined to R1=0.0962 for 360 unique reflections, with Fo>4σF and 27 refined parameters. It can be described as being formed by electroneutral [Cu3I(CH3S)2]0 {100} layers stacked along a. The origin of delchiaroite is related to the supergene alteration of enargite in vugs of Liassic marbles. Chemically, this mineral belongs to the pseudo-binary system CuI–Cu(CH3S) at a 1:2 ratio. It constitutes the first example of natural iodide–methanethiolate, illustrating the interaction between inorganic and organic chemistry. Its name honors the mineral collector Lorenzo Del Chiaro (b. 1951) for his contribution to the knowledge of the mineralogy of the vugs of the Marble Formation belonging to the Alpi Apuane metamorphic complex.
- Article
(6433 KB) - Full-text XML
-
Supplement
(202 KB) - BibTeX
- EndNote
The Apuan Alps mountain range (northern Tuscany, Italy) is well-known worldwide for the presence of the quarries of the Carrara marbles that have been exploited for more than 2000 years. Since the end of the 16th century (Del Riccio, 1597), they have also been known for the occurrence of well-crystallized mineral specimens in the rare vugs of the marbles, particularly studied since the second half of the 19th century. At the beginning of the 20th century, the number of mineral species known from the marble quarries was about a dozen, mainly thanks to the studies of Giovanni D'Achiardi (1872–1944) (e.g., D'Achiardi, 1899, 1905a, 1905b, 1906, 1911a, 1911b, 1931). However, at that time, only the most common mineral species were known, and it was only after the 1970s that the actual mineral diversity of the vugs of the Carrara marbles was revealed (e.g., Franzini et al., 1987; Orlandi and Franzini, 1994; Orlandi et al., 1996; Orlandi, 2007; Biagioni et al., 2019).
Currently, more than 120 different mineral species have been identified in the vugs of the Carrara marble. The majority of them belong to the sulfide and sulfosalt class, but several supergene minerals are also known. Among this large number of species, six have their type locality in this area, i.e., the lead sulfosalts bernarlottiite, disulfodadsonite, and moëloite (Orlandi et al., 2002, 2013, and 2017) and the supergene minerals carraraite, zaccagnaite, and zincalstibite (Merlino and Orlandi, 2001; Bonaccorsi et al., 2007). All of these new species have been described in the last 25 years, owing to the technological advancement of analytical techniques allowing for the study of very low amounts of available material. Moreover, the collaboration between mineralogists and mineral collectors was crucial in favoring the examination of a large number of specimens.
During a routine check of mineral specimens through energy-dispersive spectroscopy (EDS), some very small acicular crystals characterized by the presence of Cu, I, and S were identified. This phase was found in a specimen provided by the quarryman Paolo Cagnoni, who collected it in May 2025 during the exploitation of a marble quarry. Following these preliminary data, spectroscopic and structural investigations, along with quantitative chemical analysis, allowed for the full characterization of this mineral species that turned out to be a hitherto unknown compound.
The mineral, its name (delchiaroite), and its symbol (Dch) were approved by the Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (CNMNC-IMA) under voting no. 2025-076. The name honors Lorenzo Del Chiaro (b. 30 April 1951) for his contribution to the knowledge of the mineralogy of the cavities of the Carrara marbles. During the 1980s and 1990s, he collaborated with researchers from the University of Pisa in the study of this kind of occurrence (Orlandi and Del Chiaro, 1989; Del Chiaro and Lari, 1990), and he actively sampled the sulfosalt occurrences of the Seravezza marble basin (Orlandi et al., 1996). Del Chiaro provided the specimen of robinsonite used for the solution of its crystal structure by Franzini et al. (1992), as well as type material for the description of the new Ge mineral carraraite (Merlino and Orlandi, 2001) and of the Pb persulfosalt moëloite (Orlandi et al., 2002). He also provided several studied specimens of sulfosalts from the Buca della Vena mine (e.g., Orlandi, 2003). Finally, Del Chiaro was co-author of a book about the mineralogy of the vugs of the Carrara marble (Biagioni et al., 2019).
Type material of delchiaroite is represented by two co-type specimens. One specimen (i.e., a marble fragment with supergene minerals and a single crystal used for the X-ray diffraction study) is kept in the mineralogical collection of the Museo di Storia Naturale of the University of Pisa, Via Roma 79, Calci (Pisa, Italy), under catalogue no. 20083. The other specimen (i.e., polished grain used for chemical analysis) is stored in the National Museum in Prague under catalogue no. P1P 29/2025.
This paper describes the new mineral species delchiaroite, discusses its crystal chemistry, and puts forward some hypotheses about its genesis.
Delchiaroite was discovered in a sample collected at La Piana quarry (latitude 44°04′41′′ N, longitude 10°08′23′′ E), Colonnata marble basin, Carrara, Apuan Alps, Massa-Carrara Province, Tuscany, Italy (Fig. 1). La Piana quarry exploits the well-known Marble Formation, a Lower Liassic carbonate sequence affected by greenschist facies metamorphism (e.g., Meccheri et al., 2007) and belonging to the Apuane Unit. The latter, along with the Massa Unit, forms the so-called Alpi Apuane metamorphic complex (e.g., Carmignani and Kligfield, 1990), with two recorded main tectonic events, i.e., a first compressional ductile phase (D1) during the Oligocene–Miocene boundary (ca. 27 Ma; e.g., Kligfield et al., 1986) and a later extensional D2 phase. According to Vaselli et al. (2012), the cavities were formed during the late stages of the tectonic evolution of the Apuane Metamorphic Complex, probably after the ductile-folding phases (D1 and early D2) and before high-angle faulting events (final stages of late D2), i.e., at the ductile–brittle transition during the initial stage of late D2. Late-stage veins probably formed at T=250 °C and P=0.2 GPa (about 8 km in depth; Vaselli et al., 2012). It is probable that the mineralization continued down to very low T (probably down to ambient temperature), as exemplified by the occurrence of low-T minerals (e.g., native sulfur), in agreement with Bracci et al. (1978).

Figure 1Location of La Piana quarry. Top right: a view of La Piana quarry (modified after Google Earth, Imagery © 2025 Airbus). The red star indicates the sampling site, shown at the bottom left of the top-right photo (picture taken on 8 July 2025; photo by Cristian Biagioni).
Delchiaroite occurs as light-yellow acicular crystals, elongated on [010], up to 0.1 mm in length (Fig. 2). Its streak is light yellow, and the luster is greasy. It is transparent, and it does not fluoresce under both short- and long-wavelength ultraviolet radiations. Hardness was not determined owing to the paucity of available material, but it should be soft. Delchiaroite is brittle; scanning electron microscope observations suggest the occurrence of a perfect cleavage parallel to the crystal elongation. Calculated density, based on the ideal formula and single-crystal X-ray diffraction unit-cell parameters, is 3.565 g cm−3.

Figure 2Delchiaroite, light-yellow acicular crystals, with light-blue lavendulan and green zincolivenite on calcite. La Piana quarry, Colonnata basin, Carrara, Massa-Carrara Province, Tuscany, Italy. Holotype specimen, Natural History Museum of the University of Pisa. Catalogue no. 20083. Photo by Cristian Biagioni.
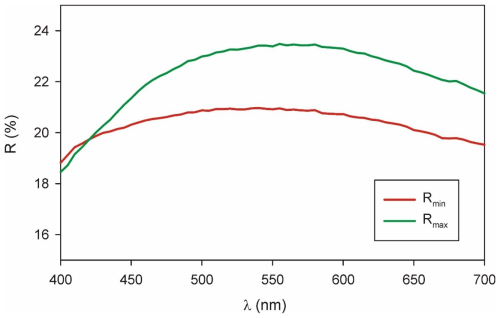
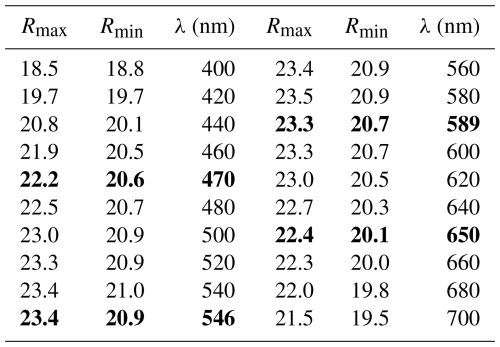
Due to the very small amount of available material of delchiaroite, the optical properties were determined on the polished grains embedded in epoxy for electron microprobe analysis, determining its properties in reflected light. Delchiaroite is, thus, light gray in color, with abundant yellow internal reflections. Pleochroism and bireflectance were not observed, maybe being masked by the strong internal reflections. This may also affect the anisotropism that is moderate, with gray tints. Reflectance values were measured in air with a spectrophotometer (Tidas MSP400) attached to a Leica microscope (100× objective), using a WTiC (Zeiss no. 370) standard. Results for the 400–700 nm range are given in Table 1 and are shown in Fig. 3. The mean refractive index of delchiaroite could not be calculated using the Gladstone–Dale relationship (Mandarino, 1976, 1981) due to the lack of the parameter for (CH3S)−.
Table 1Reflectance values (%) for delchiaroite. Values for the four COM wavelengths are shown in bold.
In type material, delchiaroite is associated with lavendulan, native copper, theisite, zincolivenite, and altered enargite. Its origin seems to be related to the supergene alteration of this latter copper sulfoarsenate.
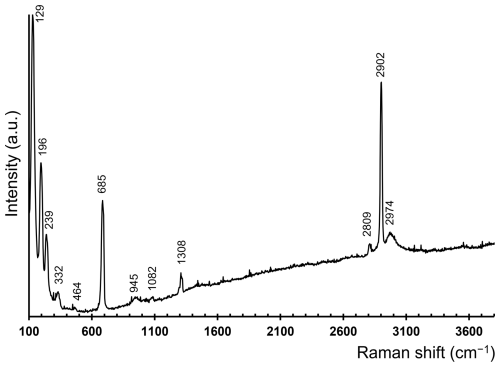
3.1 Raman spectroscopy
Micro-Raman spectra were collected on a sample of delchiaroite in the range of 100–4000 cm−1 using a Horiba Xplora apparatus mounted on a confocal Olympus BX41 microscope (Dipartimento di Scienze della Terra, University of Pisa, Italy). The Raman signal was excited by an unpolarized green-diode-pumped solid-state laser (λ=532 nm) and measured by a CCD detector. The experimental parameters were as follows: 50× objective, 60 s exposure time, three exposures, 1200 lines per mm grating, and 2.5 mW laser power level. The minimum lateral and depth resolutions were set to a few micrometers. The system was calibrated using the 520.6 cm−1 Raman band of silicon before each experimental session. The thermal damage of the measured points was excluded by visual inspection of the excited surface after measurement, by the observation of the possible decay of spectral features at the start of excitation, and by checking for thermal downshift of Raman lines.
3.2 Chemical data
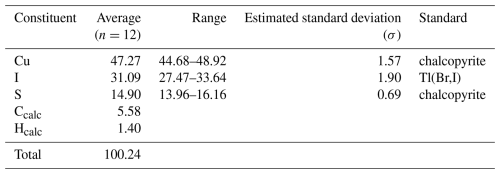
Quantitative chemical analyses were carried out using a Cameca SX 100 electron microprobe (WDS mode) on a polished crystal surface at the National Museum, Prague, Czech Republic. Delchiaroite was unstable under the electron beam, and different conditions were tried. The better experimental conditions were the following: accelerating voltage of 15 kV, beam current of 2 nA, and beam size of 2 µm. Sulfur decreased during the experiment, and we used the time-dependent measurement methodics (S is measured for a total of 20 s, in separate intervals of 4 s, and the result is recalculated to the time 0). The standards (element, emission line) were as follows: chalcopyrite (Cu Kα, S Kα) and Tl(Br,I) (I Lα). Matrix correction by means of a PAP procedure (Pouchou and Pichoir, 1985) was applied to the data. The results of the chemical analyses (average of 12 spot analyses) are given in Table 2. As there is not enough pure material available for a direct determination of C and H, their amount was calculated based upon the assumed stoichiometry.
3.3 X-ray crystallography
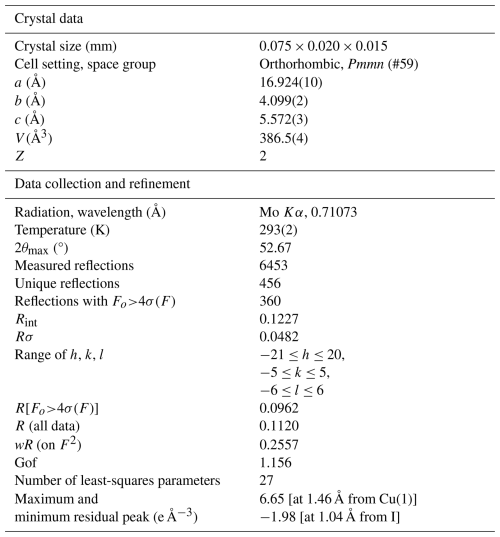
Intensity data were collected using a Bruker D8 Venture diffractometer equipped with an air-cooled Photon III CCD detector and microfocus Mo Kα radiation (CISUP, University of Pisa, Italy). The detector-to-crystal distance was 38 mm. Data were collected using ω and φ scan modes, in 0.5° slices, with an exposure time of 120 s per frame. A total of 1601 frames were collected. The frames were integrated with the Bruker SAINT software package using a narrow-frame algorithm. Data were corrected for Lorentz polarization, absorption, and background using the Apex4 software package (Bruker AXS Inc., 2022). Unit-cell parameters were refined on the basis of the XYZ centroid of 1326 reflections above 20 σ(I) with 4.813°<2θ<44.56°. They are a=16.924(10), b=4.099(2), c=5.572(3) Å, and V=386.5(4) Å3.
The statistical tests on the distribution of values ( = 0.724) suggested the absence of a center of symmetry. According to systematic absences, the crystal structure of delchiaroite was solved in some different orthorhombic space groups using Shelxtl (Sheldrick, 2015a). A solution was found in the acentric Pmn21 space group, with unit-cell parameters transformed with respect to those given above according to the matrix [0 1 0 | 0 0 1| 1 0 0]. However, the structure refinement indicated the occurrence of racemic twinning, with a twin component ratio of 0.5(5). Moreover, a check with PLATON (Spek, 2009) suggested a higher symmetry due to the occurrence of a center of symmetry. For this reason, the solution in the centric space group Pmmn was proposed, and the crystal structure was refined using Shelxl-2018 (Sheldrick, 2015b). Neutral scattering curves, taken from the International Tables for Crystallography (Wilson, 1992), were used. The structure solution allowed us to locate the position of one I atom, two Cu atoms, one S atom, and one C one. After several cycles of isotropic refinement, R1 converged to 0.1873, with high-density residuals around I. Modeling the atomic displacement parameters of Cu, S, and I as anisotropic, the R1 factor was lowered to 0.1138. Some residuals around the C atom suggested the presence of H atoms, but the CH3 group was modeled using the AFIX137 instruction (Sheldrick, 2015b). The R1 factor was slightly lowered to 0.1125. The single-crystal X-ray diffraction pattern of delchiaroite showed the contribution of several misaligned grains, and an examination of the list of the most disagreeable reflections revealed the occurrence of six reflections with higher than . After the omission of the first two most disagreeable reflections, the R1 was lowered to 0.1001, further lowered by the omission of four further reflections. In the last stage of the crystal structure refinement, the positions of H atoms were fixed because their refinement did not allow for a correct convergence of the structural model, probably due to their disordered arrangement around the C atom. After several cycles of anisotropic refinement for all atoms but the H ones, the conventional R1 factor converged to 0.0962 for 360 unique reflections with Fo>4σF and 27 refined parameters. Two relatively high residuals around Cu(1) and I were interpreted as being due to the bad quality of the diffraction data (Rint=0.1227). Even if the model is presented with H atoms, it should be noted that these latter atoms are too close to C (around 0.96 Å instead of ∼ 1.08 Å – e.g., Marynick and Dixon, 1977), resulting in overbonding of C atoms (see below). Removing H atoms, the refinement converged to R1=0.0968.
Details of the data collection and crystal structure refinement are given in Table 3. Atom coordinates and isotropic or equivalent isotropic displacement parameters are reported in Table 4, whereas Table 5 gives selected bond distances. Bond valence calculation, shown in Table 6, was performed using the bond parameters of Brese and O'Keeffe (1991).
Table 4Sites, Wyckoff positions, site occupancy (s.o.), fractional atom coordinates, and equivalent isotropic or isotropic (*) displacement parameters (in Å2) for delchiaroite.
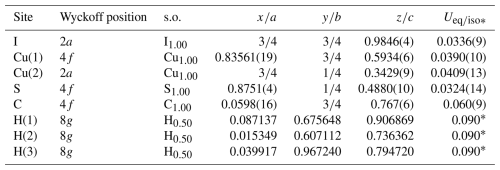
Table 6Bond valences (in v.u.) for delchiaroite.
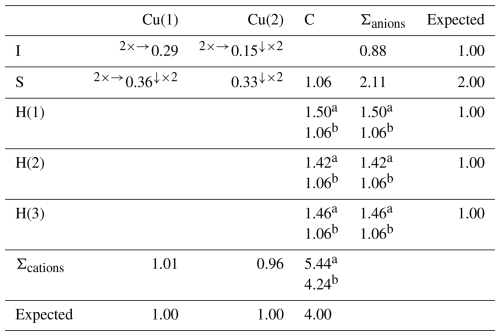
a Bond valence value calculated using the refined C–H distances; b calculated using a C–H distance of 1.079 Å, in agreement with Marynick and Dixon (1977).
Owing to the very small amount of pure material and its small size, X-ray powder diffraction data for delchiaroite were collected using a Bruker D8 Venture single-crystal diffractometer equipped with a Photon III CCD area detector (microfocus CuKα radiation) simulating a Gandolfi-like geometry. The observed X-ray diffraction lines are reported in Table 7, along with the calculated pattern based on the structural model discussed below. Unit-cell parameters, refined using the software UnitCell (Holland and Redfern, 1997) on the basis of 26 unequivocally indexed reflections, are a=16.8721(19), b=4.0992(4), c=5.5841(9) Å, and V=386.21(6) Å3.
Table 7X-ray powder diffraction data (d in Å) for delchiaroite.
Icalc and dcalc were obtained using PowderCell 2.4 (Kraus and Nolze, 1996) on the basis of the structural model of delchiaroite given in Table 4. Only calculated reflections with Icalc > 2 are reported, if not observed. The seven strongest reflections are shown in bold. a Reflection probably due to the occurrence of very minor calcite. * Reflections used for the refinement of unit-cell parameters.
4.1 Raman spectrum of delchiaroite
The Raman spectrum of delchiaroite is shown in Fig. 4. The strong band at 2902 cm−1, associated with two weaker bands at 2809 and 2974 cm−1, can be due to C–H stretching modes in CH3 groups (e.g., Allkins and Hendra, 1966). No distinct bands related to O–H modes were observed. The band at 1308 cm−1 is due to the symmetrical bending of CH3 groups, whereas weak bands at 945 and 1082 cm−1 can be related to CH3 rocking modes (e.g., Scott and McCullough, 1958; Meyer, 1992). The relatively strong band at 685 cm−1 is due to C–S single bonds (e.g., Scott and McCullough, 1958; Sexton and Nyberg, 1986; Meyer, 1992). Bands at wavenumbers lower than 500 cm−1 are probably related to Cu–S and Cu–I modes, as well as to lattice modes. Weak bands at 332 and 464 cm−1 may be due to Cu–S bonds (e.g., Mernagh and Trudu, 1993; Andrew et al., 1994). Finally, it is worth noting that the strong Raman band at 129 cm−1 reminds us of that observed at 122 cm−1 in the spectra of marshite, CuI (Lafuente et al., 2015) and in synthetic CuI (Li et al., 2022), as well as the band at 128 cm−1 reported by Welch et al. (2016) in siidraite, Pb2Cu(OH)2I3.
4.2 Chemical formula of delchiaroite
The collection of quantitative chemical data for delchiaroite by an electron microprobe was a very difficult task for several reasons. The first problem is related to the very limited amount of available material and its very small size, which made difficult the preparation of polished surfaces for chemical analyses. However, we were successful, and we obtained some very small flat surfaces on which it was possible to perform several spot analyses. Unfortunately, other issues remained. In some cases, owing to the extreme thinness of the acicular crystals of delchiaroite, the electron beam interacted with epoxy resin, thus lowering the total amount of the analyzed elements. Moreover, the mineral proved to be highly unstable under the electron beam, and several tests were conducted under various conditions, allowing us to find the better analytical conditions. However, S volatilization occurred, and a time-dependent measurement protocol was applied. Notwithstanding these issues, the agreement between the analytical ratios and those derived from the structural data, together with the Raman evidence (C–H and C–S vibrational modes), lead to the overall consistency of the results.
The empirical formula of delchiaroite, calculated on the basis of Cu + I + S = 6 atoms per formula unit, is Cu3.07(8)I1.01(7)(CH3S)1.92(8). The ideal, end-member formula is Cu3I(CH3S)2, corresponding to (in wt %) Cu 46.30, S 15.57, H 1.47, C 5.83, and I 30.82.
4.3 Crystal structure of delchiaroite
The crystal structure of delchiaroite is shown in Fig. 5. It can be described as being formed by chains of Cu(1) and S atoms, running along b and decorated by CH3 groups, giving the electroneutral formula [Cu(1)CH3S]0. Every chain is bonded to a symmetrical one along a through Cu(2) atoms, forming ribbons with the chemical composition [Cu(1)2Cu(2)(CH3S)2]1+. These ribbons are connected, along c, by rows of tetrahedrally coordinated I atoms, forming electroneutral {100} layers of composition [Cu(1)2Cu(2)I(CH3S)2]0. Finally, these layers are stacked along a, very probably through van der Waals interactions, which would permit a {100} perfect cleavage.
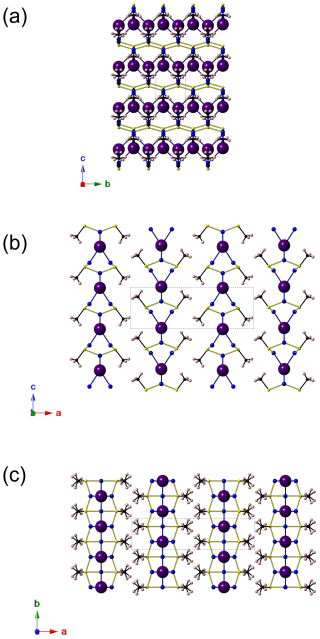
Figure 5Projection of the crystal structure of delchiaroite down a (a), b (b), and c (c), respectively. Copper, C, H, S, and I atoms are shown as blue, black, pink, yellow, and violet circles, respectively. Carbon–H bonds are shown as thick black lines, as well as C–S bonds; Cu–S bonds are shown as thick yellow lines, while Cu–I bonds are thick blue lines. Drawn with CrystalMaker (Palmer, 2014).
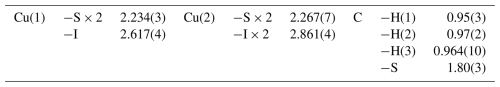
Copper is hosted at two symmetry-independent sites, namely Cu(1) and Cu(2). The former has a 3-fold, triangular coordination, forming two Cu–S (at 2.234 Å) and one Cu–I (at 2.617 Å) bonds (Fig. 6). Cu(2) displays a distorted tetrahedral coordination, with two Cu–S (at 2.267 Å) and two Cu–I (at 2.861 Å) bonds (Fig. 6). Copper–S distances agree with those observed in Cu(CH3S), ranging from 2.23 to 2.28 Å (Baumgartner et al., 1993). Relatively short Cu–Cu contacts occur, i.e., Cu(1)–Cu(2) = 2.872(3) Å and Cu(1)–Cu(1) = 2.898 Å; these values can be compared with that reported by Welch et al. (2016) in siidraite, Pb2Cu(OH)2I3, i.e., 2.782 Å. The bond valence sums at Cu(1) and Cu(2) are 1.01 and 0.96 valence units (v.u.), in agreement with the occurrence of Cu+ at both sites.
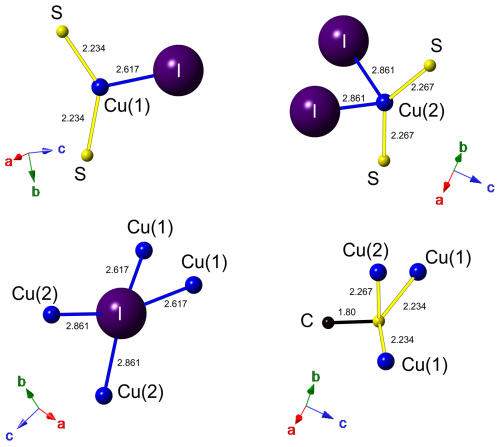
Figure 6Coordination environment for atoms hosted at Cu(1), Cu(2), I, and S sites. Same symbols as in Fig. 5. Drawn with CrystalMaker (Palmer, 2014).
Carbon is hosted at the C site and is bonded to three H atoms and one S atom at 1.80 Å. In synthetic Cu(CH3S), the C–S distance was fixed at 1.835 Å (Baumgartner et al., 1993). Carbon–H distances (∼ 0.96 Å) are too short with respect to the ideal one of ∼ 1.08 Å (e.g., Marynick and Dixon, 1977) reported in CH3 groups. This results in a severe overbonding of C (5.44 v.u.); on the contrary, by using an ideal C–H distance of 1.08 Å, the bond valence sum at the C atom is 4.24 v.u., in agreement with the presence of C4+.
Iodine is tetrahedrally coordinated by Cu atoms hosted at two Cu(1) sites and two Cu(2) sites (Fig. 6), with an average bond distance of 2.739 Å. Its bond valence sum is 0.88 v.u. In marshite, the observed Cu–I distance is 2.625 Å (Cooper and Hawthorne, 1997), whereas Welch et al. (2016) found average < Cu–I > distances of 2.655 and 2.634 Å in siidraite.
Sulfur is bonded to three Cu atoms, i.e., two at Cu(1) and one at Cu(2), and to one C atom in a distorted S(Cu3C) tetrahedral arrangement (Fig. 6), similarly to what was observed in synthetic Cu(CH3S) by Baumgartner et al. (1993). As stated above, interlayer van der Waals bonding probably occurs between {100} layers, as suggested by HS distances (3.11 Å) slightly longer than the sum of the van der Waals radii of these two chemical species (3.00 Å; e.g., Fargher et al., 2022).
Taking into account site multiplicity, the formula of delchiaroite obtained through the crystal structure refinement is Cu3I(CH3S)2 (Z=2). This formula can be compared with the results of chemical analysis. The atomic ratio – according to the structural investigation – is in agreement with chemical data, giving the ratio .
4.4 Genesis of delchiaroite
A supergene origin of the delchiaroite-bearing assemblage also involving native copper, zincolivenite, lavendulan, and theisite is suggested, being formed at the expense of altered enargite. Supergene minerals found in the cavities of the Marble Formation originated through the interaction between primary sulfides and low-T meteoric fluids whose percolation is favored by widespread karstic phenomena. Following Pekov et al. (2011), low-T conditions are necessary to form iodide species (T<50–70 °C) because, at higher T, iodine is highly mobile. The coexistence of native copper, along with Cu2+-bearing lavendulan, zincolivenite, and theisite, as a result of the alteration of Cu+-bearing enargite indicates peculiar redox conditions. In fact, delchiaroite is a Cu+-bearing phase.
It is also worth noting that there is the occurrence of a Cl-bearing phase (lavendulan) in close association with delchiaroite, an indication of the circulation of halogen-bearing fluids. Our new evidence is not a single anomaly as four halogen-bearing species are currently known from the vugs of the Marble formation at Carrara: connellite, lavendulan, mimetite, and vanadinite (Biagioni et al., 2019, and references therein). These species contain Cl, whereas delchiaroite is the first I-bearing species.
Dealing with the possible origin of I, S, and organic components constituting delchiaroite, both intraformational and extraformational sources could have been involved based on the existing evidence.
The Carrara marble is characterized by the smell of rotten eggs when broken. Usually, such a smell is interpreted as being due to the occurrence of H2S. This is supported by high sulfate concentrations measured in leachates of fluid inclusions hosted by the Carrara marble, as reported by Prochaska (2023), which may result from oxidation of H2S (or other reduced-S species) by atmospheric O2 during the preparation of the leachate. In addition, the halogen systematics of these leachates indicate extraordinary enrichment of included fluids in both Br and I (Fig. 7). This feature is likely to be related to thermal conversion of organic matter into graphite (present in some marble horizons) during regional metamorphism, which was associated with mobilization of I and Br (originally bound to organic matter) into metamorphic fluids. The host Carrara marble can thus represent a fertile local source of both I and S. On the contrary, it is probable that the contribution of a thermally immature external source is necessary for the methanethiolate anion because aliphatic thiols decompose at temperatures above ca. 170–250 °C (Yang et al., 2006), and the peak temperatures of the metamorphic overprint of the Carrara marble were between 350 and 450 °C (e.g., Carmignani and Kligfield, 1990; Molli et al., 2000, 2002).
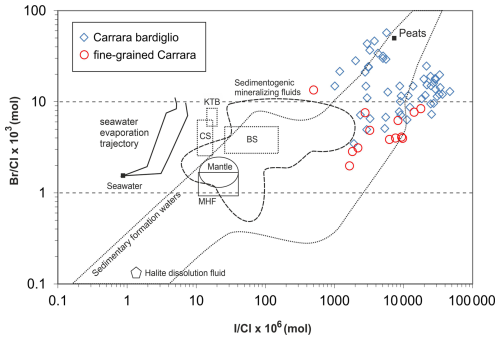
Figure 7 vs. plot of fluid inclusion leachates from the Carrara marbles (data after Prochaska, 2023) and their comparison with fluids of various origins. Abbreviations: MHF – uncontaminated magmatic hydrothermal fluids; CS – shield brines from the Canadian Shield; BS – shield brines from the Baltic Shield; KTB – deep saline fluids drilled by the KTB borehole (Bavaria, Bohemian Massif). References for comparative data are listed in Dolníček et al. (2009).
Three potential sources of methanethiolate can be suggested in the study area. The first one can be represented by sedimentary rocks of the Tuscan Nappe, a tectonic unit overlying the Carrara marble. A hydraulic connection of both of these formations was already suggested by Costagliola et al. (1999) to explain the high salinity of late-stage fluid inclusions in hydrothermal veins hosted in the Carrara marble. Furthermore, the participation of late fluids dominated by meteoric waters during the formation of hydrothermal veins in Carrara marble (Costagliola et al., 1999; Molli et al., 2010) gives evidence for the long-lasting existence of migration paths from the surface into the marble body and supports the possibility of the transfer of some components leached from overlying sediments.
The second potential source of organic thiols could be represented by organic-rich soil layers (e.g., Rao et al., 2014) as these still cover marble outcrops outside the quarry (Fig. 1). Moreover, organic-rich soils can retain and store iodine as organo-iodine compounds (e.g., Keppler et al., 2004). These chemical species may then be leached by meteoric waters, percolate along karstified fractures, and interact with weathering enargite.
The third possible source may be found in marine aerosols in rain. The Apuan Alps rise abruptly from the narrow coastal plain bordering the Ligurian Sea and are characterized by high precipitation (>2500 mm yr−1; Rapetti and Vittorini, 1994). Oceanic phytoplankton is the source of dimethyl sulfide (CH3SCH3), which is one of the most important sulfur species released into the atmosphere (e.g., Martínez et al., 2000). However, it is probable that this trace chemical species would be largely oxidized during atmospheric transport, thus barely representing the actual source for the crystallization of delchiaroite. Nevertheless, the marine aerosols may have contributed both Cl and I (e.g., Sturges and Barrie, 1988); marine water as a possible source of halogens in the genesis of halides has been proposed by other authors (e.g., Whiterhead, 1919; Andreu et al., 2015).
In summary, available evidence supports the possibility of local sources for I, S, and hydrocarbon compounds. However, as delchiaroite is a very late-stage mineral, formed though supergene processes, the choice of a unique source for iodine and organic compounds is not straightforward, and further research is necessary.
A final consideration is as follows: iodides rapidly decompose under the effect of sunlight and atmospheric O2 (e.g., Pekov et al., 2011). The specimen where delchiaroite was identified was collected by the quarryman Paolo Cagnoni during an exploitation activity and was not found in the quarry dump, where the mineral may have rapidly decomposed and/or oxidized. It is possible that this immediate sample collection after the extraction could have allowed for the preservation of delchiaroite. Considering the enrichment in I of the Carrara marble shown by the results of Prochaska (2023), as well as their possible ephemeral nature, the presence of so-far unrecognized iodide minerals among secondary mineral assemblages at Carrara cannot be excluded.
Table 8Organic Cu-bearing minerals.
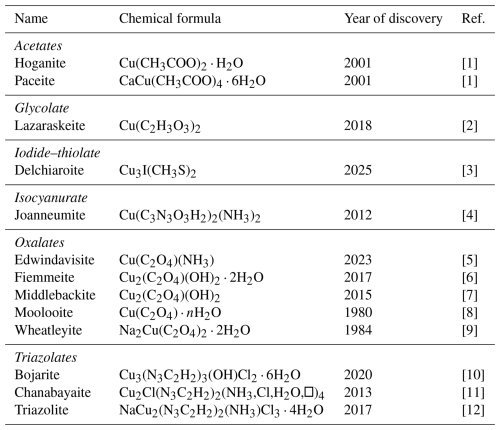
[1] Hibbs et al. (2002). [2] Yang et al. (2022). [3] This work. [4] Bojar et al. (2017). [5] Yang et al. (2025). [6] Demartin et al. (2018). [7] Elliott (2019). [8] Clarke and Williams (1986). [9] Rouse et al. (1986). [10] Chukanov et al. (2020). [11] Chukanov et al. (2015). [12] Chukanov et al. (2018).
4.5 Relations with other species
Natural iodide–chalcogenides are known, i.e., demicheleite-(I), BiSI (Demartin et al., 2010); hanauerite, AgHgSI (Pekov et al., 2023); mutnovskite, Pb2AsS3(I,Cl,Br) (Zelenski et al., 2006); perroudite, Hg5Ag4S5(I,Br)2Cl2 (Sarp et al., 1987); and radtkeite, Hg3S2ICl (McCormack et al., 1991). Moreover, two copper iodides are known, namely marshite, CuI (Cooper and Hawthorne, 1997), and siidraite, Pb2Cu(OH)2I3 (Rumsey et al., 2017).
Copper is also involved in the crystallization of several organic minerals (Table 8). Among the 12 currently known species, 10 have been described in the last 25 years, and 8 were found after 2010. However, none of them contain S as an essential constituent. Indeed, delchiaroite is the 13th organic mineral containing Cu, and it is the first natural Cu iodide–organochalcogenide, showing a novel crystal structure. Synthetic halo-thio-organometallic compounds are actively studied for their luminescence properties (e.g., Knorr et al., 2014; Rajput et al., 2015).
In addition, the synthetic phase [Cu(CH3S)]∞ is known (Baumgartner et al., 1993). It was obtained through thermal decomposition of [C3H7N][Cu4(CH3S)]6 ⋅ CH4O, whereas other thiolates described by Baumgartner et al. (1993) were obtained from solutions kept at 55 °C for 1 h and then evaporated at room temperature. Thus, all of these compounds seem to be low-T phases. The synthetic Cu(CH3S) phase has a unit-cell volume of V=285.95 Å3 (Z=4). Delchiaroite, Cu3I(CH3S)2, can be considered to be a combination of CuI + 2 Cu(CH3S). Copper iodide is known as the mineral marshite, having a unit-cell volume of V=221.45 Å3 (Z=4). Thus, the volume Vmix of CuI + 2Cu(CH3S) would be Å3. Delchiaroite has Z=2, and, thus, the volume of this mineral (i.e., 386.5 Å3) has to be compared with 2Vmix=396.7 Å3.
The discovery of delchiaroite is a further confirmation of the results of Grew et al. (2017) regarding the less-than-expected predictive power of synthetic compounds; indeed, notwithstanding the synthesis of some copper thiolates (e.g., Baumgartner et al., 1993), the complexity of natural environments and geological processes is able to add some further chemical constituents, as represented, in this case, by iodine.
Thus, delchiaroite is the first natural iodide–methanethiolate mineral and shows a novel crystal structure among mineral species. Further studies focusing on its synthesis under controlled conditions would be useful in understanding the low-T processes occurring in the Earth's critical zone, where the lithosphere interacts with the atmosphere, hydrosphere, and biosphere. Indeed, the description of delchiaroite may represent a new step in our understanding of the complex interaction between inorganic and organic chemistry, and, as the (CH3S)− groups probably have a biological origin, the identification of this iodide–chalcogenide mineral may be considered to be a kind of biosignature.
The Crystallographic Information File data of delchiaroite are available in the Supplement.
The supplement related to this article is available online at https://doi.org/10.5194/ejm-38-153-2026-supplement.
CB collected the preliminary data. CB and AC carried out the field study. CB collected the micro-Raman spectrum, and JS and ZD performed the electron microprobe analysis and collected the optical data. CB and YM critically examined the data.
At least one of the (co-)authors is a member of the editorial board of European Journal of Mineralogy. The peer-review process was guided by an independent editor, and the authors also have no other competing interests to declare.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
Paolo Cagnoni is warmly acknowledged for providing us with the specimen of delchiaroite used for the mineralogical investigation. The comments by Nikita V. Chukanov and an anonymous reviewer improved the original paper.
This research was supported by the Ministry of Culture of the Czech Republic (long-term project DKRVO 2024-2028/1.II.c; National Museum, grant no. 00023272) for Jiří Sejkora and Zdeněk Dolníček.
This paper was edited by Sergey Krivovichev and reviewed by Nikita V. Chukanov and one anonymous referee.
Allkins, J. R. and Hendra, P. J.: The infra-red and Raman spectra of dimethyl sulphide, selenide and telluride, Spectrochim. Acta, 22, 2075–2081, 1966.
Andreu, E., Torró, L., Proenza, J. A., Domenech, C., García-Casco, A., Villanova de Benavent, C., Chavez, C., Espaillat, J., and Lewis, J. F.: Weathering profile of the Cerro de Maimón VMS deposit (Dominican Republic): textures, mineralogy, gossan evolution and mobility of gold and silver, Ore Geol. Rev., 65, 165–179, 2015.
Andrew, C. R., Yeorn, H., Selverstone Valentine, J., Karlsson, B. G., Bonander, N., van Pouderoyen, G., Canters, G. W., Loehr, T. M., and Sanders-Loehr, J.: Raman spectroscopy as an indicator of Cu–S bond length in Type 1 and Type 2 copper cysteinate proteins, J. Am. Chem. Soc., 116, 11489–11498, 1994.
Baumgartner, M., Schmalle, H. W., and Baerlocher, C.: Synthesis, characterization, and crystal structure of three homoleptic copper(I) thiolates: (Cu(CH3S−))∞, [(C6H5)4P+]2[Cu5(CH3S−)7] ⋅ C2H6O2, and [(C3H7)4N+]2[Cu4(CH3S)6 ⋅ CH4O, J. Solid St. Chem., 107, 63–75, 1993.
Biagioni, C., Orlandi, P., Camarda, S., Chinellato, M., Appiani, R., Del Chiaro, L., and Sanguineti, G.: Minerals from marbles of Carrara and the Apuan Alps, LoGisma Editore, Florence, 144 pp., ISBN: 978-88-94926-15-6, 2019.
Bojar, H., Walter, F., and Baumgartner, J.: Joanneumite, Cu(C3N3O3H2)2(NH3)2, a new mineral from Pabellón de Pica, Chile and the crystal structure of its synthetic analogue, Mineral. Mag., 81, 155–166, 2017.
Bonaccorsi, E., Merlino, S., and Orlandi, P.: Zincalstibite, a new mineral, and cualstibite: crystal chemical and structural relationships, Am. Mineral., 92, 198–203, 2007.
Bracci, G., Dalena, D., and Orlandi, P.: I geodi del marmo di Carrara, Atti Soc. Tosc. Sci. Nat., Mem., 85, 221–241, 1978.
Brese, N. E. and O'Keeffe, M.: Bond-valence parameters for solids, Acta Crystallogr., B47, 192–197, 1991.
Bruker AXS Inc.: APEX4, Bruker Advanced X–ray Solutions, Madison, Wisconsin, USA, 2022.
Carmignani, L. and Kligfield, R.: Crustal extension in the Northern Apennines: the transition from compression to extension in the Alpi Apuane core complex, Tectonics, 9, 1275–1303, 1990.
Chukanov, N. V., Zubkova, N. V., Möhn, G., Pekov, I. V., Pushcharovsky, D. Y., and Zadov, A. E.: Chanabayaite, Cu2(N3C2H2)2Cl(NH3,Cl,H2O,□)4, a new mineral containing triazolate anion, Zap. Ross. Mineral. Obsh., 144, 36–47, 2015 (in Russian).
Chukanov, N. V., Zubkova, N. V., Möhn, G., Pekov, I. V., Belakovskiy, D. I., Van, K. V., Britvin, S. N., and Pushcharovsky, D. Y.: Triazolite, NaCu2(N3C2H2)2(NH3)2Cl3 ⋅ 4H2O, a new mineral species containing 1,2,4-triazolate anion, from a guano deposit at Pabellón de Pica, Iquique Province, Chile, Mineral. Mag., 82, 1007–1014, 2018.
Chukanov, N. V., Möhn, G., Zubkova, N. V., Ksenofontov, D. A., Pekov. I. V., Agakhanov, A. A., Britvin, S. N., and Desor, J.: Bojarite, Cu3(N3C2H2)3(OH)Cl2 ⋅ 6H2O, a new mineral species with a microporous metal–organic framework from the guano deposit at Pabellón de Pica, Iquique Province, Chile, Mineral. Mag., 84, 921–927, 2020.
Clarke, R. M. and Williams, I. R.: Moolooite, a naturally occurring hydrated copper oxalate from Western Australia, Mineral. Mag., 50, 295–298, 1986.
Cooper, M. A. and Hawthorne, F. C.: A note on the crystal structure of marshite, Can. Mineral., 35, 785–786, 1997.
Costagliola, P., Benvenuti, M., Maineri, C., Lattanzi, P., and Ruggieri, G.: Fluid circulation in the Apuane Alps core complex: evidence from extension veins in the Carrara marble, Mineral. Mag., 63, 111–122, 1999.
D'Achiardi, G.: Minerali dei marmi di Carrara (Nota preventiva), Atti Soc. Tosc. Sci. Nat., Proc. Verb., 11, 160–163, 1899.
D'Achiardi, G.: I minerali dei marmi di Carrara, Atti Soc. Tosc. Sci. Nat., Mem., 21, 47–57, 1905a.
D'Achiardi, G.: I minerali dei marmi di Carrara, Atti Soc. Tosc. Sci. Nat., Mem., 21, 236–264, 1905b.
D'Achiardi, G.: I minerali dei marmi di Carrara (Aggiunte alle parti I e II), Atti Soc. Tosc. Sci. Nat., Proc. Verb., 15, 47–48, 1906.
D'Achiardi, G.: Minerali dei marmi di Carrara, Atti Soc. Tosc. Sci. Nat., Proc. Verb., 20, 54–58, 1911a.
D'Achiardi, G.: Minerali dei marmi di Carrara, Atti Soc. Tosc. Sci. Nat., Proc. Verb., 20, 77–78, 1911b.
D'Achiardi, G.: Fluorite nei marmi dell'Orto di Donna, Period. Mineral., 2, 141, 1931.
Del Chiaro, L. and Lari, A.: Osservazioni sui solfuri dei marmi delle Apuane, Riv. Mineral. Ital., 13, 171–174, 1990.
Del Riccio, A.: Istoria delle pietre, Umberto Allemandi, Torino, 256 pp. (reprint), 1597.
Demartin, F., Gramaccioli, C. M., and Campostrini, I.: Demicheleite-(I), BiSI, a new mineral from La Fossa Crater, Vulcano, Aeolian Islands, Italy, Mineral. Mag., 74, 141–145, 2010.
Demartin, F., Campostrini, I., Ferretti, P., and Rocchetti, I.: Fiemmeite Cu2(C2O4)(OH)2 ⋅ 2H2O, a new mineral from Val di Fiemme, Trentino, Italy, Minerals, 8, 248, https://doi.org/10.3390/min8060248, 2018.
Dolníček, Z., Fojt, B., Prochaska, W., Kučera, J., and Sulovský, P.: Origin of the Zálesí U–Ni–Co–As–Ag/Bi deposit, Bohemian Massif, Czech Republic: fluid inclusion and stable isotope constraints, Miner. Deposita, 44, 81–97, 2009.
Elliott, P.: Middlebackite, a new Cu oxalate mineral from Iron Monarch, South Australia: Description and crystal structure, Mineral. Mag., 83, 427–433, 2019.
Fargher, H. A., Sherbow, T.J., Haley, M.M., Johnson, D.W., and Pluth, M.D.: C–HS hydrogen bonding interactions, Chem. Soc. Rev., 51, 1454–1469, 2022.
Franzini, M., Orlandi, P., Bracci, G., and Dalena, D.: Minerals of the Carrara marble, Miner. Rec., 18, 263–296, 1987.
Franzini, M., Orlandi, P., and Pasero, M.: Morphological, chemical and structural study of robinsonite (Pb4Sb6S13) from Alpi Apuane, Italy, Acta Vulcanol., 2, 231–235, 1992.
Grew, E. S., Hystad, G., Hazen, R. M., Krivovichev, S. V., and Gorelova, L. A.: How many boron minerals occur in Earth's upper crust?, Am. Mineral., 102, 1573–1587, 2017.
Hibbs, D. E., Kolitsch, U., Leverett, P., Sharpe, J. L., and Williams P. A.: Hoganite and paceite, two new acetate minerals from the Potosi mine, Broken Hill, Australia, Mineral. Mag., 66, 459–464, 2002.
Holland, T. J. B. and Redfern, S. A. T.: UNITCELL: a nonlinear least-squares program for cell-parameter refinement and implementing regression and deletion diagnostics, J. Appl. Crystallogr., 30, 84–84, 1997.
Keppler, F., Biester, H., Putschew, A., Silk, P. J., Schöler, H. F., and Müller, G.: Organoiodide formation during humification in peatlands, Environ. Chem. Lett., 1, 219–233, 2004.
Kligfield, R., Hunziker, J., Dallmeyer, R. D., and Schamel, S.: Dating of deformation phases using K-Ar and techniques; results from the Northern Apennines, J. Struct. Geol., 7, 781–798, 1986.
Knorr, M., Khatyr, A., Dini Aleo, A., El Yaagoubi, A., Strohmann, C., Kubicki, M. M., Rousselin, Y., Aly, S. M., Fortin, D., Lapprand, A., and Harvey, P. D.: Copper(I) halides (X = Br, I) coordinated to bis(arylthiol)methane ligands: aryl substitution and halide effects on the dimensionality, cluster size, and luminescence properties of the coordination polymers, Cryst. Growth Des., 14, 5373–5387, 2014.
Kraus, W. and Nolze, G.: PowderCell – a program for the representation and manipulation of crystal structures and calculation of the resulting X-ray powder patterns, J. Appl. Crystallogr., 29, 301–303, 1996.
Lafuente, B., Downs, R. T., Yang, H., and Stone, N.: The power of databases: the RRUFF project, in: Highlights in Mineralogical Crystallography, edited by: Armbruster, T. and Danisi, R. M., Berlin, Germany, W. De Gruyter, 1–30, https://doi.org/10.1515/9783110417104-003, 2015.
Li, Z. H., He, J. X., Lv, X. H., Chi, L. F., Egbo, K. O., Li, M. D., Tanaka, T., Guo, Q. X., Yu, K. M., and Liu, C. P.: Optoelectronic properties and ultrafast carrier dynamics of copper iodide thin films, Nat. Commun., 13, 6346, https://doi.org/10.1038/s41467-022-34117-8, 2022.
Mandarino, J. A.: The Gladstone-Dale relationship – Part I: derivation of new constants, Can. Mineral., 14, 498–502, 1976.
Mandarino, J. A.: The Gladstone-Dale relationship: part IV. The compatibility concept and its application, Can. Mineral., 19, 441–450, 1981.
Martínez, E., Albaladejo, J., Notario, A., and Jiménez, E.: A study of the atmospheric reaction of CH3S with O3 as a function of temperature, Atmosph. Environ., 34, 5295–5302, 2000.
Marynick, D. S. and Dixon, D. A.: Electron affinity of the methyl radical: Structures of CH3 and CH, P. Natl. Acad. Sci. USA, 74, 410–413, 1977.
McCormack, J. K., Dickson, F. W., and Leshendok, M. P.: Radtkeite, Hg3S2ClI, a new mineral from the McDermitt mercury deposit, Humboldt County, Nevada, Am. Mineral., 76, 1715–1721, 1991.
Meccheri, M., Molli, G., Conti, P., Blasi, P., and Vaselli, L.: The Carrara Marbles (Alpi Apuane, Italy): a geological and economical updated review, Z. Deutsch. Gesell. Geowissen., 158, 719–735, 2007.
Merlino, S. and Orlandi, P.: Carraraite and zaccagnaite, two new minerals from the Carrara marble quarries: their chemical compositions, physical properties, and structural features, Am. Mineral., 86, 1293–1301, 2001.
Mernagh, T. P. and Trudu, A. G.: A laser Raman microprobe study of some geologically important sulphide minerals, Chem. Geol., 103, 113–127, 1993.
Meyer, M.: Infrared, Raman, microwave and ab initio study of dimethyl disulfide: structure and force field, J. Molec. Struct., 273, 99–121, 1992.
Molli, G., Conti, P., Giorgetti, G., Meccheri, M., and Oesterling, N.: Microfabric studies on the deformational and thermal history of the Alpi Apuane marbles (Carrara marbles), Italy, J. Struct. Geol., 22, 1809–1825, 2000.
Molli, G., Giorgetti, G., and Meccheri, M.: Tectono-metamorphic evolution of the Alpi Apuane Metamorphic Complex: new data and constraints for geodynamic models, Boll. Soc. Geol. It., 1, 789–800, 2002.
Molli, G., Cortecci, G., Vaselli, L., Ottria, G., Cortopassi, A., Dinelli, E., Mussi, M., and Barbieri, M.: Fault zone structure and fluid-rock interaction in a high angle normal fault in Carrara marble (NW Tuscany, Italy), J. Struct. Geol., 32, 1334–1348, 2010.
Orlandi, P.: Buca della Vena: i solfosali aciculari di piombo e antimonio, Riv. Mineral. Ital., 27, 224–229, 2003.
Orlandi, P.: Seit über 2000 Jahren Marmorabbau in der Toskana. Geologie und Mineralien der Apuaner Alpen. Die Mineralien der Marmorgebiete rund um Carrara, bei Massa und der Versilia, Lapis, 7-8/2007, 15–58, 2007.
Orlandi, P. and Del Chiaro, L.: Nuovi dati sui minerali delle geodi del marmo di Carrara, Atti Soc. Tosc. Sci. Nat., Mem., 96, 313–325, 1989.
Orlandi, P. and Franzini, M.: I minerali del marmo di Carrara, Silvana Editoriale, Milan, 110 pp., ISBN 88-366-0485-4, 1994.
Orlandi, P., Del Chiaro, L., and Pagano, R.: Minerals of the Seravezza marble, Tuscany, Italy, Min. Rec., 27, 47–58, 1996.
Orlandi, P., Meerschaut, A., Palvadeau, P., and Merlino, S.: Lead-antimony sulfosalts from Tuscany (Italy). V. Definition and crystal structure of moëloite, Pb6Sb6S14(S3), a new mineral from the Ceragiola marble quarry, Eur. J. Mineral., 14, 599–606, 2002.
Orlandi, P., Biagioni, C., Moëlo, Y., and Bonaccorsi, E.: Lead-antimony sulfosalts from Tuscany (Italy). XIV. Disulfodadsonite, Pb11Sb13S30(S2)0.5, a new mineral from the Ceragiola marble quarry, Apuan Alps: occurrence and crystal structure, Eur. J. Mineral., 25, 1005–1016, 2013.
Orlandi, P., Biagioni, C., Bonaccorsi, E., Moëlo, Y., and Paar, W. H.: Lead-antimony sulfosalts from Tuscany (Italy). XXI. Bernarlottiite, Pb12(As10Sb6)Σ16S36, a new N = 3.5 member of the sartorite homologous series from the Ceragiola marble quarry: occurrence and crystal structure, Eur. J. Mineral., 29, 713–726, 2017.
Palmer, D. C.: CrystalMaker, CrystalMaker Software Ltd., Begbroke, Oxfordshire, England, 2014.
Pekov, I. V., Lykova, I. S., Bryzgalov, I. A., Ksenofontov, D. A., Zyryanova, L. A., and Litvinov, N. D.: Uniquely high-grade iodide mineralization in the oxidation zone of the Rubtsovskoe base-metal deposit, northwest Altai, Russia, Geol. Ore Dep., 53, 683–698, 2011.
Pekov, I. V., Zubkova, N. V., Britvin, S. N., Agakhanov, A. A., Polekhovsky, Y. S., Pushcharovsky, D. Y., Möhn, G., Desor, J., and Blass, G.: A new mineral hanauerite, AgHgSI, and common crystal chemical features of natural mercury sulphohalides, Crystals, 13, 18, https://doi.org/10.3390/cryst13081218, 2023.
Pouchou, J. L. and Pichoir, F.: “PAP” (φρZ) procedure for improved quantitative microanalysis, in: Microbeam Analysis, edited by: Armstrong, J. T., San Francisco Press, San Francisco, 104–106, 1985.
Prochaska, W.: Trading marble for steel: early Roman import of Carrara marble into the Alps–The example of the Magdalensberg trading post in Noricum, Minerals, 13, 1036, https//doi.org/10.3390/min13081036, 2023.
Rajput, G., Kumar Yadar, M., Drew M. G. B., and Singh, N.: Impact of ligand framework on the crystal structures and luminescent properties of Cu(I) and Ag(I) clusters and coordination polymer derived from thiolate/iodide/dppm ligands, Inorg. Chem., 54, 2572–2579, 2015.
Rao, B., Simpson, C., Lin, H., Liang, L., and Gu, B.: Determination of thiol functional groups on bacteria and natural organic matter in environmental systems, Talanta, 119, 240–247, 2014.
Rapetti, F. and Vittorini, S.: Carta climatica della Toscana centro-settentrionale, Pacini Editore, Pisa, 1994.
Rouse, R. C., Peacor, D. R., Dunn, P. J., Simmons, W. B., and Newbury, D.: Wheatleyite, Na2Cu(C2O4)2 ⋅ 2H2O, a natural sodium copper salt of oxalic acid, Am. Mineral., 71, 1240–1242, 1986.
Rumsey, M. S., Welch, M. D., Kleppe, A. K., and Spratt, J.: Siidraite, Pb2Cu(OH)2I3, from Broken Hill, New South Wales, Australia: the third halocuprate(I) mineral, Eur. J. Mineral., 29, 1027–1030, 2017.
Sarp, H., Birch, W. D., Hlava, P. F., Pring, A., Sewell, D. K. B., and Nickel, E. H.: Perroudite, a new sulfide-halide of Hg and Ag from Cap-Garonne, Var, France, and from Broken Hill, New South Wales, and Coppin Pool, Western Australia, Am. Mineral., 72, 1251–1256, 1987.
Scott, D. W. and McCullough, J. P.: Characteristic vibrational frequencies of organic sulfur compounds, J. Am. Chem. Soc., 80, 3554–3558, 1958.
Sexton, B. A. and Nyberg, G. L.: A vibrational and TDS study of sulfur adsorbates on Cu(100): evidence for CH3S species, Surf. Sci., 165, 251–267, 1986.
Sheldrick, G. M.: SHELXT – Integrated space-group and crystal-structure determination, Acta Crystallogr., A71, 3–8, 2015a.
Sheldrick, G. M.: Crystal structure refinement with SHELXL, Acta Crystallogr., C71, 3–8, 2015b.
Spek, A. L.: Structure validation in chemical crystallography, Acta Crystallogr., D65, 148–155, 2009.
Sturges, W. T. and Barrie, L. A.: Chlorine, bromine and iodine in Arctic aerosols, Atmosph. Environ., 22, 1179–1194, 1988.
Vaselli, L., Cortecci, G., Tonarini, S., Ottria, G., and Mussi, M.: Conditions for veining and origin of mineralizing fluids in the Alpi Apuane (NW Tuscany, Italy): Evidence from structural and geochemical analyses on calcite veins hosted in the Carrara marbles, J. Struct. Geol., 44, 76–92, 2012.
Welch, M. D., Rumsey, M. S., and Kleppe, A. K.: A naturally-occurring new lead-based halocuprate(I), J. Solid St. Chem., 238, 9–14, 2016.
Wilson, A. J. C. (Ed.): International Tables for Crystallography Volume C: Mathematical, Physical and Chemical Tables, Kluwer Academic Publishers, Dordrecht, the Netherlands, 1992.
Whiterhead, W. L.: The veins of Chañarcillo, Chile, Econ. Geol., 14, 1–45, 1919.
Yang, B., Tian, S., and Zhao S.: A study of thermal decomposition of alkanethiols in pressure reactor, Fuel Process. Technol., 87, 673–678, 2006.
Yang, H., Gu, X., Gibbs, R. B., Evans, S. H., Downs, R. T., and Jibrin, Z.: Lazaraskeite, Cu(C2H3O3)2, the first organic mineral containing glycolate, from the Santa Catalina Mountains, Tucson, Arizona, U.S.A., Am. Mineral., 107, 509–516, 2022.
Yang, H., Gu, X., Kampf, A. R., Marty, J., Gibbs, R. B., and Downs, R. T.: Edwindavisite, Cu(C2O4)(NH3), a new oxalate mineral, from the Rowley mine, Maricopa County, Arizona, USA, Mineral. Mag., 89, 535–543, 2025.
Zelenski, M., Balić-Žunić, T., Bindi, L., Garavelli, A., Makovicky, E., Pinto, D., and Vurro, F.: First occurrence of iodine in natural sulfosalts: The case of mutnovskite, Pb2AsS3(I,Cl,Br), a new mineral from the Mutnovsky volcano, Kamchatka Peninsula, Russian Federation, Am. Mineral., 91, 21–28, 2006.